What is the Einstein Refrigerator and how does it work?
Albert Einstein, the man famous for unlocking the laws of the universe, once tried his hand at engineering. The result was the Einstein refrigerator, which was patented in 1930.
Einstein designed the refrigerator in a joint effort with his former student Leó Szilárd. Szilárd went on to be an accomplished physicist in his own right and was the first to theorise atomic chain reactions. Even so, it is easy to be overshadowed in history books when your work mate was Einstein.
The impetus for the design was the danger of household refrigerators of the time. They used toxic gases and often leaked after mechanical components broke. One tragic instance where a whole family died is said to have spurred the super duo into action.
Einstein and Szilárd’s solution was a form of absorption refrigerator. These, unlike typical refrigerators of the time (and of now), do not need a mechanical compressor. They instead use a heat source and a complex interplay between substances to draw heat from one place to another. This cuts the chance of them breaking and releasing harmful gases.
The Einstein Refrigerator - key principles
Einstein may well have been one of the smartest people to have lived. That does not mean his concepts are beyond the grasp of us intellectual mortals. The Einstein refrigerator is actually quite simple – maybe beautifully so. Much like the physics work Einstein is famous for.
The Einstein refrigerator works on a couple of key principles:
When a liquid evaporates into a gas, it absorbs heat from its surroundings and produces cooling. The opposite is true too – when a gas condenses into a liquid, it releases heat into its surroundings. This heat that is transferred during the phase change of substances is called latent heat.
Partial pressure is the hypothetical pressure a gas will have when other gases it shares a container with are removed. When the partial pressure of a gas falls below its vaporisation pressure, evaporation to form more gas can occur. Likewise, when the partial pressure increases over its vaporisation pressure, condensation will occur. Altering the makeup of gases in a container by adding or removing them changes the partial pressure of existing gases.
How the Einstein Refrigerator works - schematic steps
Now that you understand the basic physics behind the Einstein Refrigerator, the steps of the cycle can be followed in the animated schematic:
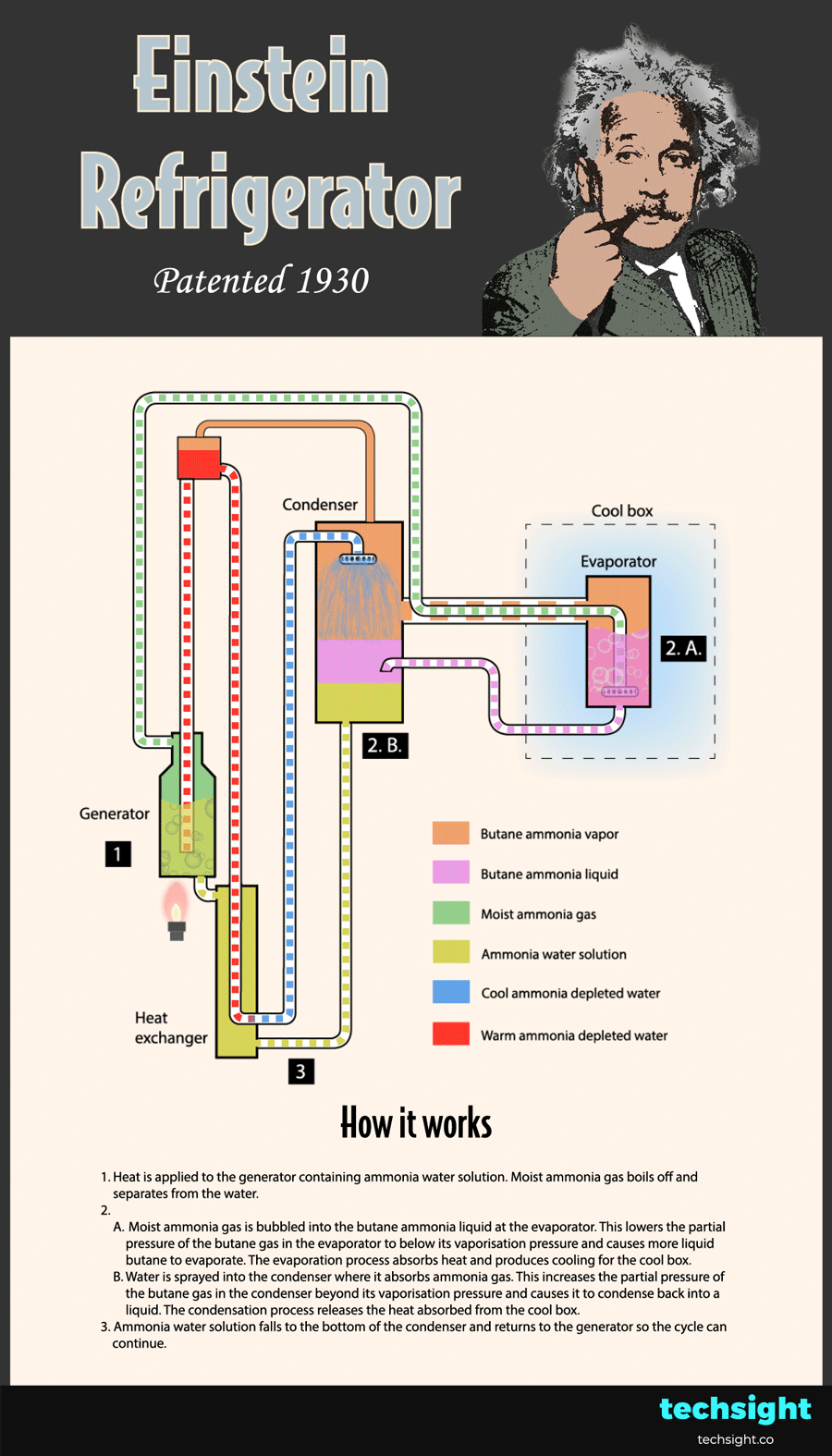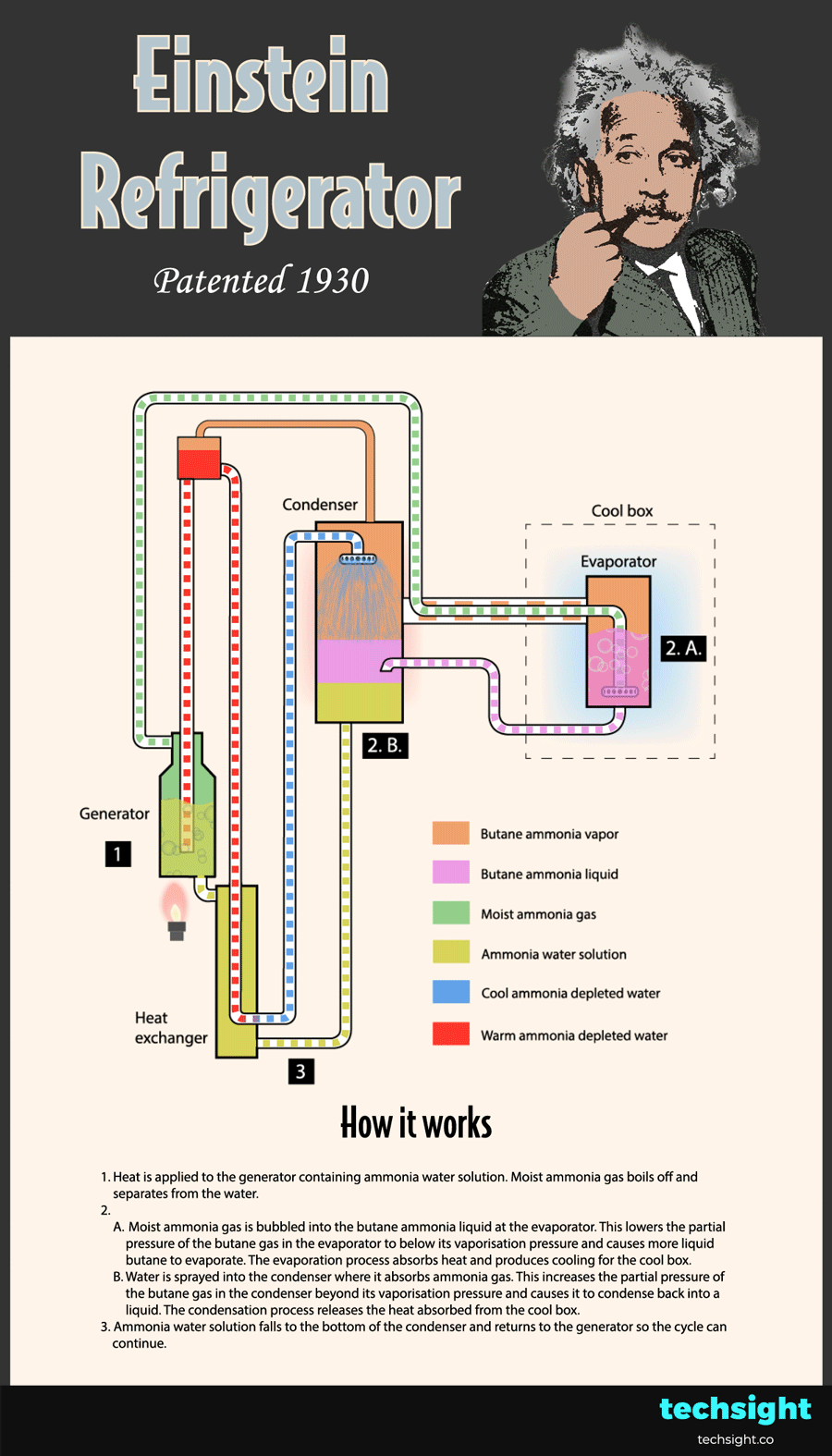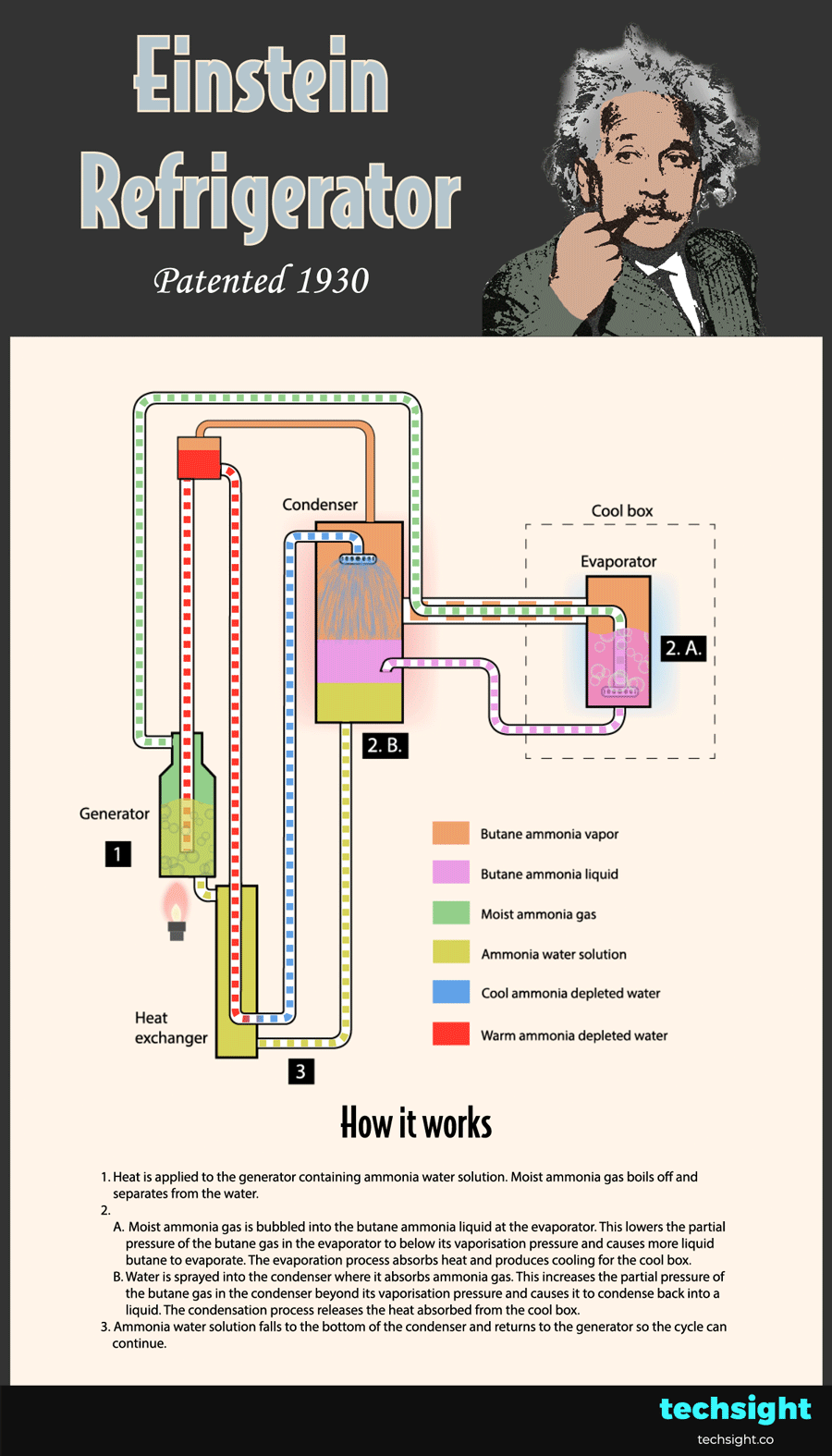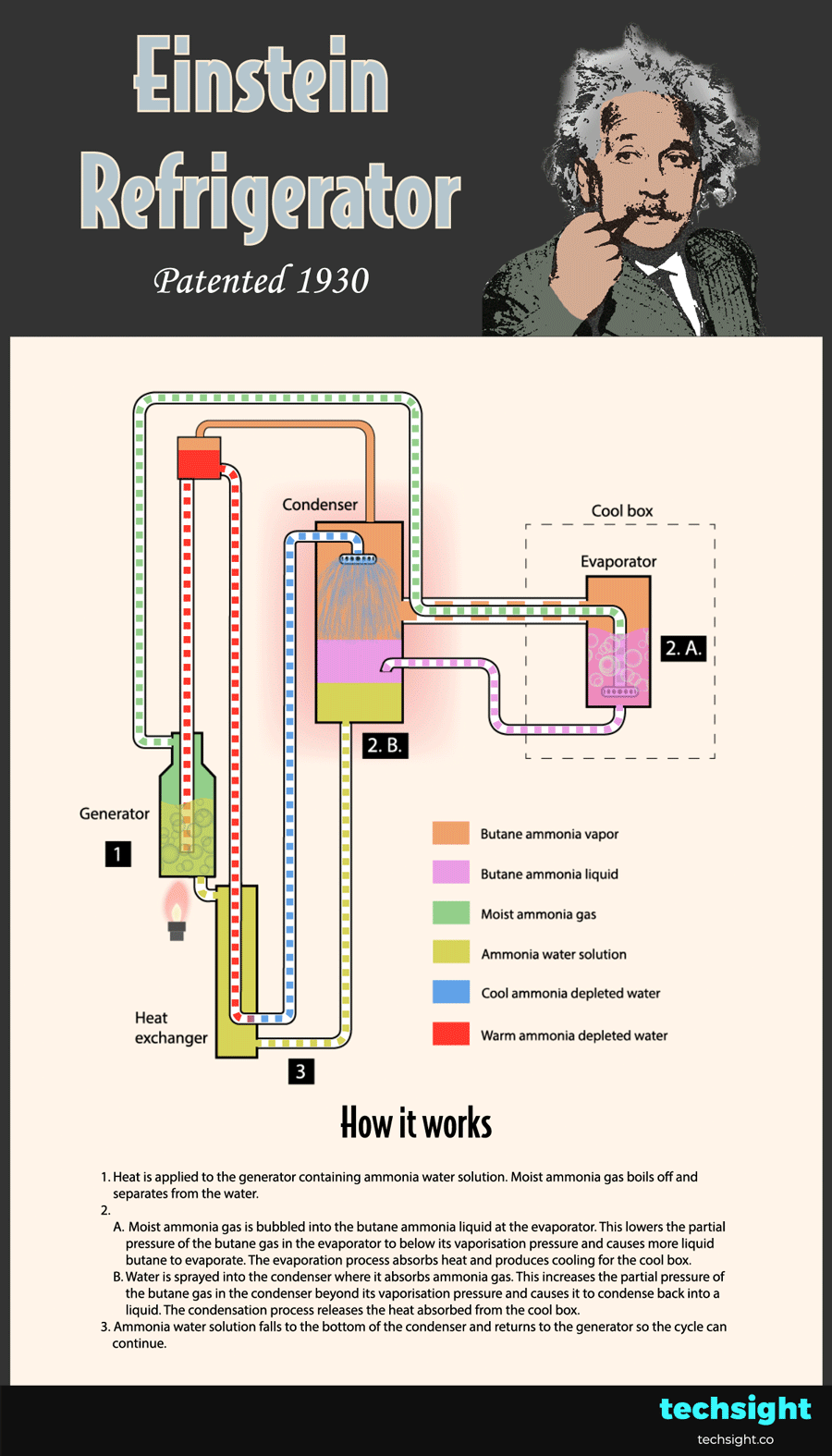
1. Heat is applied to the generator containing ammonia water solution. Moist ammonia gas boils off and separates from the water.
2.A. Moist ammonia gas is bubbled into the butane ammonia liquid at the evaporator. This lowers the partial pressure of the butane gas in the evaporator to below its vaporisation pressure. As a result, more liquid butane evaporates and absorbs heat to produce cooling for the cool box.
2.B. Water is sprayed into the condenser where it absorbs ammonia gas. This increases the partial pressure of the butane gas in the condenser beyond its vaporisation pressure. As a result, the butane gas condenses and releases the heat absorbed from the cool box.
3. Ammonia water solution falls to the bottom of the condenser and returns to the generator so the cycle can continue.
What happened to the Einstein Refrigerator? And could it make a comeback?
Despite its clever design and creators, the Einstein refrigerator was not a commercial success. This was partly because of a lack of funding, but in the end, the launch of non-toxic synthetic refrigerants eclipsed the concept.
Ironically, the world is now turning its back on modern refrigerants because of their high Global Warming Potential (GWP). Governments are bringing in tough limits that will see natural refrigerants, like ammonia, make a comeback as they contribute far less to global warming. For example, R410a which is a common refrigerant today, has a GWP of 2088 – meaning it is 2088 times more potent than CO2 for global warming. Ammonia, on the other hand, has a GWP of zero.
So maybe Einstein and Szilárd were just ahead of their time – by 100 years, to almost be exact.